Introduction
Maintaining good intestinal health is directly related to optimal productive performance. The integrity of the intestinal epithelium, associated with microbiota balance and proper digestive functionality, is critical for efficient nutrient absorption, adequate growth, and overall animal health. Furthermore, the intestine not only plays a fundamental role in digestion but also constitutes one of the first lines of defense against external pathogens, representing a key component of the organism’s immune barrier.
On the other hand, animals are primarily exposed to mycotoxins through contaminated feed, placing the gastrointestinal tract on the front line of contact with these compounds. In addition, the intestine is a primary target organ for several of these toxins, including deoxynivalenol (DON) and T-2 toxin, which belong to the trichothecene group produced by fungi of the Fusarium genus.
DON, also known as vomitoxin, is a type B trichothecene that severely disrupts the digestive system, causing vomiting, diarrhea, and feed refusal. T-2 toxin, a type A trichothecene, exhibits similar gastrointestinal toxicity and additionally induces skin lesions characterized by necrosis and inflammation. Both mycotoxins are associated with hepatic toxicity and are particularly detrimental on the intestinal epithelium, compromising this fundamental barrier that protects the organism. Consequently, they reduce nutrient absorption efficiency, increase inflammation, and impair immune function.
To address these challenges in animal nutrition, various strategies have been developed, including approaches focused on mycotoxin adsorption to the mitigation of their secondary effects. In this context, BIŌNTE® QUIMITŌX® PLUS stands out as a comprehensive solution, based on three modes of action: adsorption, bioprotection, and postbiotic effects. It represents an alternative that reduces mycotoxin bioavailability and mitigates their toxicity, thereby contributing to enhanced digestive health, optimized nutrient uptake, and healthier growth in swine.
Against this background, a study was conducted in collaboration with Schothorst Feed Research (SFR) in the Netherlands to evaluate the efficacy of BIŌNTE® QUIMITŌX® PLUS in mitigating the intestinal toxicity of DON and T-2 toxin using an innovative ex vivo model with porcine ileal organoids (IPOs).
Ex vivo model: innovation in the evaluation of anti-mycotoxin agents
An ex vivo study is a scientific experiment conducted outside a living organism, but using freshly isolated tissues, organs, or cells that still retain some of their natural functions. This design allows for:
- Obtaining realistic results without the complexity of working with the whole organism.
- Achieving greater clarity in understanding the functions of specific organs or tissues.
- Ensuring results are obtained from an ethical perspective.
Such studies represent an innovative tool for experimentation in animal production, as they allow the assessment of the toxic effects of certain substances directly on target systems, while emphasizing animal welfare and evaluating the efficacy of applied interventions.
Consequently, the objective of this study was to evaluate the efficacy of BIŌNTE® QUIMITŌX® PLUS in mitigating the negative effects induced by exposure to DON and T-2 mycotoxins in the porcine gastrointestinal tract, through the measurement of cell viability, assessed via mitochondrial activity, and intestinal permeability (Figure 1).
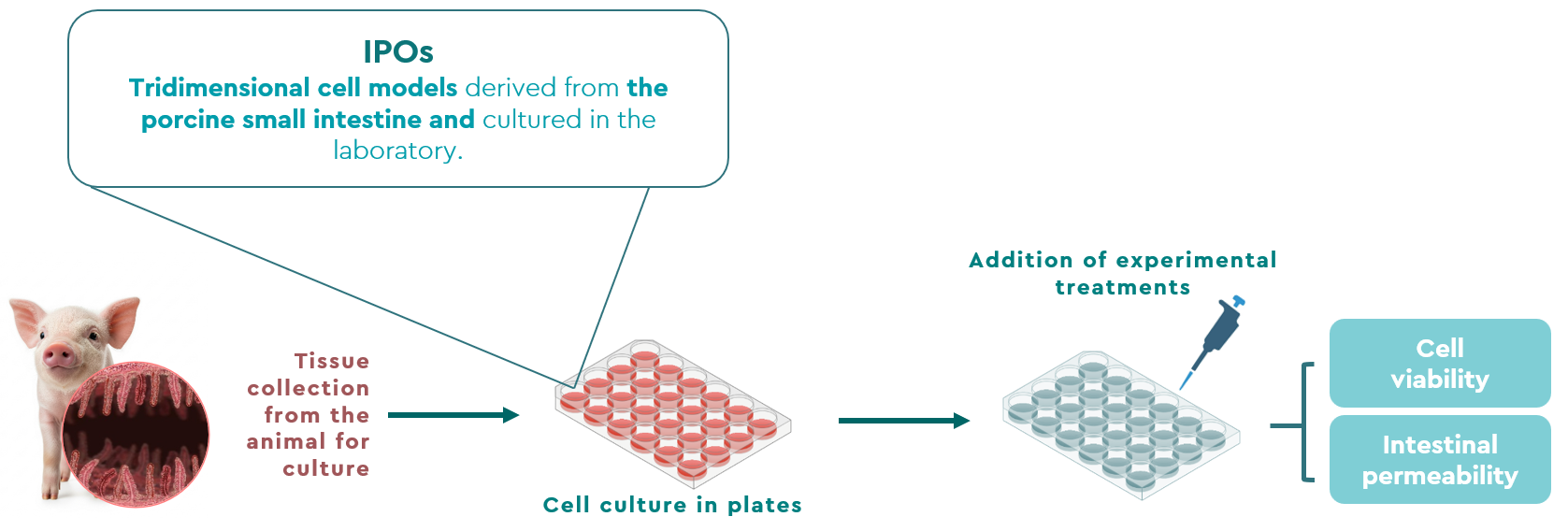
Figure 1. Schematic of the study methodology.
The study included four experimental groups, consisting of a control treatment, a treatment with the inclusion of mycotoxins (DON and T-2), a treatment with BIŌNTE® QUIMITŌX® PLUS, and a treatment combining mycotoxins and BIŌNTE® QUIMITŌX® PLUS (Table 1).
| Experimental treatments applied to the IPOs | ||
|---|---|---|
| Treatment | Ex vivo conditions | Field conditions* |
| 1. Control | – | – |
2. Mycotoxins (MYC) DON T-2 | 1 µM 0.1 µM | 296 ppb 46.7 ppb |
| 3. BIŌNTE® QUIMITŌX® PLUS (BQP) | 0.5 mg/mL | 1 kg/t |
| 4. MYC + BQP | group 2 and 3 | group 2 and 3 |
Table 1. Experimental treatments applied to the IPOs. *Theoretical extrapolation of ex vivo units to field units. They do not represent direct usage recommendations.
BIŌNTE® QUIMITŌX® PLUS increases cell viability in the porcine intestinal epithelium
Cell viability is a parameter that indicates the percentage of “alive”, biologically active cells, in a sample. Therefore, it serves as a key indicator of intestinal health and nutrient absorption capacity. A higher number of functional intestinal cells contributes to greater productive efficiency, supporting optimal growth and development. To assess intestinal cell viability, mitochondrial activity is analyzed, as it defines cell vitality and, consequently, whether the cell thrives or deteriorates.
When mitochondrial function is normal, the cell efficiently produces energy and withstands stress, thereby maintaining the integrity and functionality of the tissues. In contrast, if this activity is disrupted by exposure to toxins or other stressors, cell viability decreases, affecting the structure and function of vital tissues and organs. This, in turn, triggers inflammation and immunosuppression, resulting in reduced productive performance and an increased susceptibility to diseases.
In this ex vivo study, the effect of BIŌNTE® QUIMITŌX® PLUS on porcine intestinal cells viability was evaluated, specifically through the experimentation with the control treatment (1) and the tissue treated with BIŌNTE® QUIMITŌX® PLUS (3).
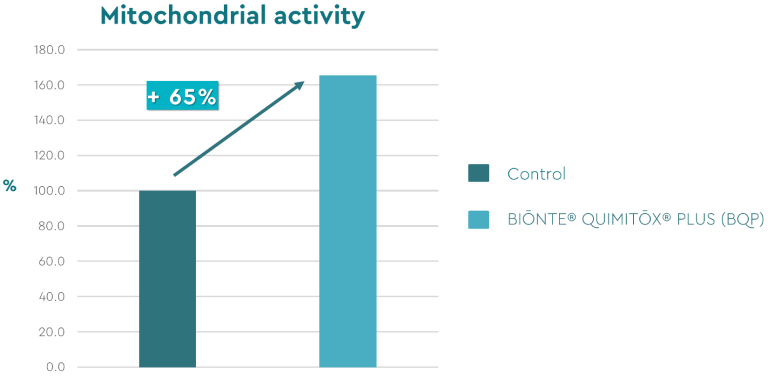
Figure 2. Mitochondrial activity results (%).
The results showed a 65% increase in mitochondrial activity in the experimental tissue following the administration of the product (Figure 2). These findings demonstrate that BIŌNTE® QUIMITŌX® PLUS enhances cell viability, thereby improving the overall health of the intestinal epithelium.
BIŌNTE® QUIMITŌX® PLUS restores the integrity of intestinal cells altered by DON and T-2 toxin exposure in swine
A healthy intestine is essential for animal health, as it functions as a selective barrier that allows the absorption of essential nutrients while preventing the translocation of bacteria, toxins, and other harmful substances into the bloodstream. However, exposure to mycotoxins such as DON and T-2 can compromise this structural integrity, resulting in a leaky gut that permits pathogenic agents to enter the organism, triggering inflammation and immunosuppression (Li et al., 2022) (Figure 3).
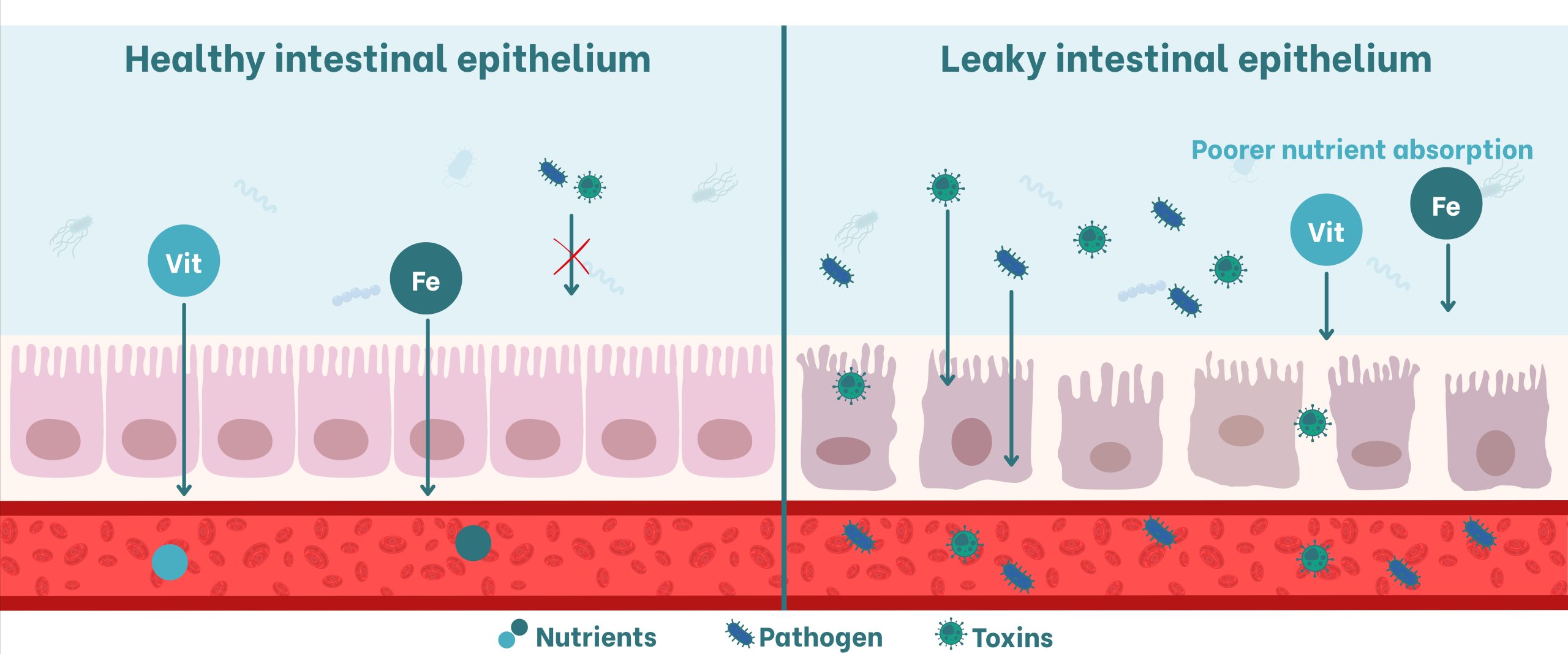
Figure 3. Schematic of intestinal permeability in response to toxins and pathogens. On the left, healthy impermeable epithelium; on the right, altered epithelium permeable to toxins and pathogens.
The consequences of altered intestinal permeability directly impact the profitability and health of swine, manifesting as weight loss, diarrhea, and low production efficiency (Wellington et al., 2020; Zhang et al., 2024). Moreover, this vulnerable state not only affects overall productivity but also significantly increases susceptibility to infections, compromising the animal’s integrity (Lessard et al., 2015).
In the present ex vivo study, intestinal permeability was evaluated to assess both: the toxic effects of mycotoxins on the intestinal epithelium and the ability of BIŌNTE® QUIMITŌX® PLUS to counteract this toxicity.
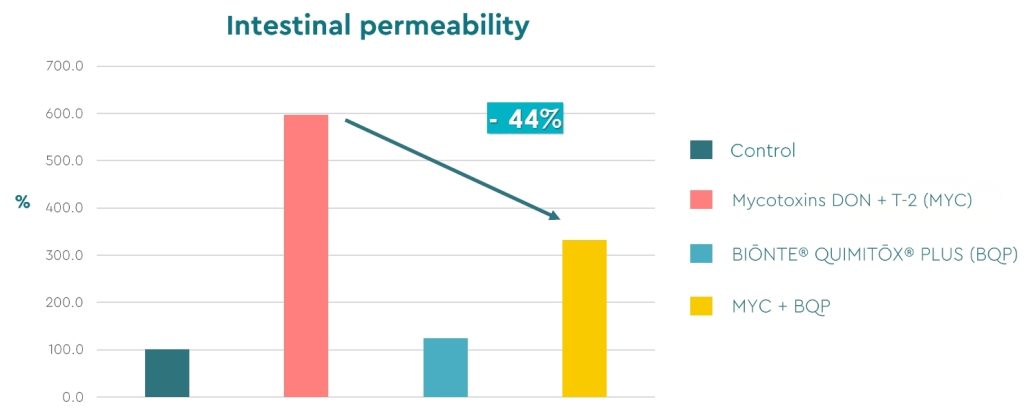
Figure 4. Intestinal permeability results (%).
The results revealed physiological values of intestinal permeability, not altered, both in the control group and in the group that received only BIŌNTE® QUIMITŌX® PLUS. Subsequently, treatment with the mycotoxins DON and T-2 caused a significant increase in permeability, compromising tissue integrity. Clinically, this alteration would be reflected in problems with nutrient absorption and reduced performance in the affected swine. Finally, the combined treatment, in which the tissue was exposed to the mycotoxins and also received BIŌNTE® QUIMITŌX® PLUS, demonstrated the efficacy of the product, showing a 44% reduction in intestinal permeability compared to tissue exposed only to DON and T-2.
Conclusion
In conclusion, animal health and performance critically depend on the integrity and proper functioning of the intestinal barrier. BIŌNTE® QUIMITŌX® PLUS protects intestinal function and shields swine from mycotoxin-induced toxicity, such as that caused by DON and T-2, contributing to reduced inflammation, enhanced immunity, and maximized performance. In this regard, studies using ex vivo models represent an ethical and innovative alternative for evaluating the toxicity of these compounds and the efficacy of interventions in animal nutrition.


