Results show the mitigation of risks from aflatoxin M1, guaranteeing dairy safety.
Introduction
The contamination of food and feed with mycotoxins represents a global threat to food safety with high economic and public health significance. This scenario drives the European Food Safety Authority (EFSA) to continuously update its risk assessments to establish maximum permitted levels in the food chain (Bodas et al., 2023). These substances are secondary metabolites naturally produced by filamentous phytopathogenic fungi, mainly from the genera Fusarium, Aspergillus, and Penicillium spp., which are chemically very stable and persistent (Schrenk et al., 2020).
The exposure of ruminants to aflatoxin B1 (AFB1) occurs through the ingestion of contaminated forage and balanced feed (Pichardo-Matamoros & Elizondo-Salazar, 2020). Cereals and their by-products represent a more significant source and risk of exposure, notably corn, wheat, barley, oats, sorghum, and rice, as well as the use of oilseeds such as soy, cotton, and peanuts (Bodas et al., 2023). Corn, for example, is the cereal with the highest prevalence of aflatoxins worldwide, and its high inclusion in dairy feed is a critical risk factor (Tolosa et al., 2021). Contamination in these raw materials is strongly favored in hot and humid climates, aggravated by drought stress in plants, in the field, or by poor storage conditions (Pichardo-Matamoros & Elizondo-Salazar, 2020).
Upon ingestion of AFB1, the rumen microbiota degrades a small fraction of AFB1 into less toxic metabolites such as aflatoxicol. Although the latter is traditionally considered less toxic, its importance lies in the fact that it can act as a reservoir, being capable of converting back into AFB1 under the action of an enzyme, which prolongs the animal’s systemic exposure (Tolosa et al., 2021). The remaining AFB1 is absorbed in the digestive tract and, through the cytochrome P450 enzyme system in the liver, is transformed via hydroxylation into aflatoxin M1 (AFM1), which is finally excreted in milk, urine, and feces (Pichardo-Matamoros & Elizondo-Salazar, 2020). AFM1 is highly water-soluble, allowing it to easily reach the mammary gland (Medina, 2023).
The transfer rate (percentage of consumed AFB1 secreted as AFM1) in ruminants generally fluctuates between 0.3% and 6.2% and can even exceed 7% (Bodas et al., 2023; Pichardo-Matamoros & Elizondo-Salazar, 2020; Medina, 2023). This rate is highly correlated with milk production volume; that is, high-producing cows or those in the early or middle stage of lactation transfer a bigger amount of toxin into the milk (Fels-Klerx & Camenzuli, 2016; Medina, 2023). Due to the very fast metabolism, AFM1 is detected in blood plasma just 5 minutes after AFB1 consumption (Gallo et al., 2008). Its excretion in milk begins to be evident between 12 and 24 hours after the consumption of the contaminated food. If ingestion is maintained continuously, the level of AFM1 in milk increases rapidly until reaching a steady state between 24 hours and 7 days of constant exposure (Diaz et al., 2004). Upon interrupting the supply of contaminated food, AFM1 concentrations drop, such that residues in milk disappear within a period of 72 to 96 hours (Pichardo-Matamoros & Elizondo-Salazar, 2020).
Due to the impact on public health, the presence of AFM1 is strictly regulated, although the maximum permitted limits vary by region and their food safety policies (Turna et al., 2022). The European Union (EFSA) applies one of the most rigorous criteria, establishing a maximum limit of 0.05 ppb (µg/kg) for raw milk, heat-treated milk, and milk intended for the manufacturing of dairy products (Tolosa et al., 2021). In contrast, other regions such as the United States (FDA) and several Latin American countries allow a limit of up to 0.5 ppb (µg/kg). Correspondingly, the maximum limit for total aflatoxin in raw materials is 20 ppb (µg/kg).
Unlike other contaminants, AFM1 possesses high thermal stability, implying that it is not inactivated by traditional methods of pasteurization, sterilization, or UHT treatment applied in the dairy industry (Medina, 2023). During processing, it is predominantly associated with casein, causing its concentration to rise 3 to more than 5 times in cheeses compared to the liquid milk used for their production (Silanikove et al., 2010).
This metabolic process entails consequences, causing severe liver damage, rumen alterations, reproductive problems, and marked immunosuppression that reduces productive performance (Tolosa et al., 2021). These effects are often confused with common herd infections or metabolic alterations.
Given this outlook, biotechnological solutions such as BIŌNTE® QUIMITOX® PLUS are required, designed under strict scientific standards whose efficacy lies in a unique combination of minerals, phytogenics, and organic components. The selected minerals act in the digestive tract by capturing AFB1 molecules before their absorption, thus preventing them from reaching the liver to be transformed by the cytochrome P450 enzyme system. Complementarily, phytogenic extracts such as silymarin protect hepatic integrity during hydroxylation processes. By drastically reducing the aflatoxin load that reaches the bloodstream, the product manages to block the final transfer of AFM1 to the mammary gland, ensuring that the milk produced meets the safety standards required by the EFSA.
To validate its efficacy in reducing the concentration of AFM1 in the milk of dairy cows, a field study was conducted in Mexico, in collaboration with the National Autonomous University of Mexico (UNAM).
Experimental design
The study was structured under a challenge model in real production conditions. The experimental design consisted of a group of 170 dairy cows from the AGRASISA farm, which were subjected to a critical profile of natural co-contamination in their diet. The ration presented high levels of aflatoxins and deoxynivalenol and low levels of zearalenone. The intervention protocol was based on the supply of a dose of 40 g/cow/day of BIŌNTE® QUIMITŌX® PLUS incorporated into the feed.
During the four weeks the study lasted, the parameters evaluated included the weekly kinetic measurement of AFM1 concentration in milk (expressed in ppb) to quantify the residual transfer rate, as well as the monitoring of metabolic integrity and animal resilience against the multi-key challenge.
Results
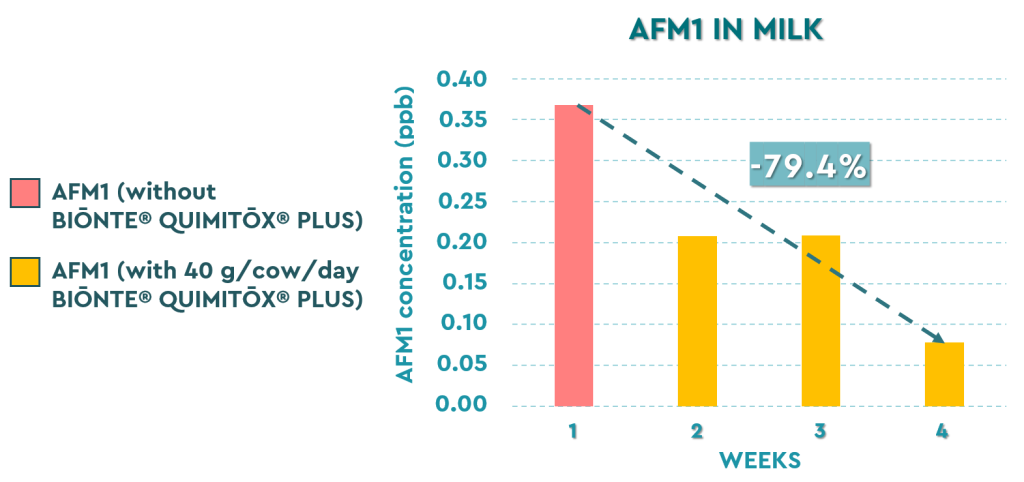
Figure 1. Decrease in AFM1 levels in milk during a 3-week period after supplying BIŌNTE® QUIMITŌX® PLUS.
The inclusion of BIŌNTE® QUIMITOX® PLUS in the animals’ diet demonstrated a drastic and sustained reduction of the toxin in the final product. In the first week of initial control, the AFM1 concentration in milk was approximately 0.37 ppb. After starting supplementation, a significant decrease was observed during weeks two and three, reaching its point of maximum remission in the fourth week with levels of approximately 0.08 ppb. At the end of the study, BIŌNTE® QUIMITOX® PLUS demonstrated its high capacity for adsorption and bioprotection by achieving a total reduction of 79.4% in the concentration of AFM1 in milk compared to the initial values
Conclusion
BIŌNTE® QUIMITOX® PLUS is a robust and effective solution for mitigating the excretion of toxic residues in cows exposed to multi-contaminated diets, ensuring both the health and longevity of the herd, as well as the food safety required by global markets and public health.


