Introduction
Toxicokinetics refers to the study of the processes by which potentially toxic substances are handled in the body (EFSA). This involves an understanding of the absorption, distribution, metabolism and excretion (ADME) of such substances. Investigating these processes across different animal species is essential, as is assessing exposure to individual mycotoxins in relation to their specific toxicological properties.
Aflatoxins
Chemical properties
Aflatoxins are characterised by a chemical structure composed of four aromatic rings, resulting in a rigid and planar molecular conformation. The most relevant mycotoxins within this group are aflatoxins B1, B2, G1 and G2, as well as aflatoxin M1, the latter being a metabolite of aflatoxin B1. B and G aflatoxins differ in the structure of their six-membered lactone ring, while the distinction between the 1 and 2 forms is based on the oxidation of the double bond. In addition to these compounds, other aflatoxin metabolites have been identified, including aflatoxicol, which is formed through the reduction of the ketone group; aflatoxin P1 (AFP1), in which the ether group is converted into an alcohol; and aflatoxin Q1 (AFQ1), which contains an alcohol group in the cyclopentanone moiety.
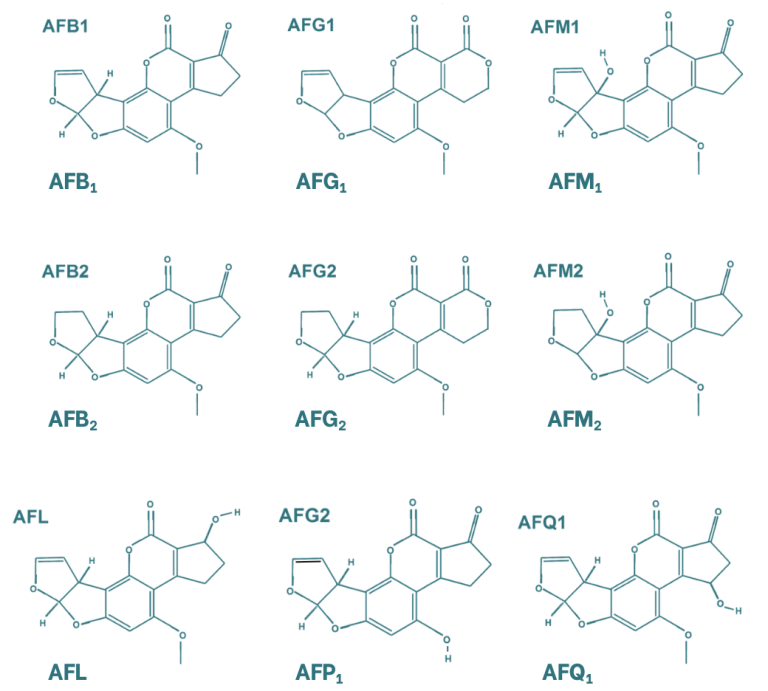
Figure 1. Chemical structure of the main aflatoxins and their metabolites (Popescu et al., 2022).
Absorption
The absorption of aflatoxin B1 (AFB1) occurs mainly in the small intestine and is largely influenced by its molecular weight and lipophilic nature (Schrenk et al., 2020a).
Distribution
The liver is the primary organ affected by aflatoxins. However, these mycotoxins can also accumulate in muscle tissue (Popescu et al., 2022).
Metabolism
AFB1 is metabolised during its passage through the gastrointestinal tract; however, the precise site at which the most extensive metabolism occurs has not yet been clearly established. Hepatic exposure to this toxin largely depends on the concentration of AFB1, the rate of intestinal absorption, and the portal hepatic blood flow (Schrenk et al., 2020a).
In the liver, aflatoxins act as substrates for CYP monooxygenases, including CYP3A4, CYP3A5, and CYP1A2. A key step in the bioactivation of AFB1 is the formation of AFB1-exo-8,9-epoxide (AFBO), its reactive metabolite.
Furthermore, AFB1 can be converted into several metabolites via different metabolic reactions. One of these metabolites is aflatoxicol, which is formed in the liver through the reduction of AFB1 mediated by an NADPH-dependent reductase. Other metabolites include aflatoxin M1 (AFM1) and aflatoxin Q1 (AFQ1), which are formed through hydroxylation; and aflatoxin P1 (AFP1), which is generated by demethylation.
An important enzymatic process involved in reducing the reactive form of AFB1 to a less reactive metabolite, such as AFB1-dialcohol, is mediated by the family of NADPH-dependent aldo-keto-reductases. The metabolites AFM1, AFP1, AFQ1 and aflatoxin-dialcohol can subsequently be conjugated with glucuronic acid and excreted in the faeces or urine. In contrast to AFB1, information on the metabolism of other aflatoxins is limited, with only scarce literature available on the metabolism of AFB2 (Schrenk et al., 2020a).
Excretion
The excretion of AFB1 and its metabolites occurs mainly via the biliary route, followed by urinary excretion. In lactating females, a proportion of the ingested AFB1 is also excreted in milk, primarily in the form of aflatoxin M1 (AFM1), together with other metabolites (Eaton et al., 1994).
Deoxynivalenol
Chemical properties
Deoxynivalenol (DON) is a mycotoxin belonging to the trichothecene family, characterised by a common structure consisting of three rings and an epoxide group at C-12. Within this group, four types are recognised (A, B, C, and D), with types A and B being the most frequently found in nature. DON is classified as a type B trichothecene and is distinguished by the presence of a ketone group at C-8 position.
Figure 2. Chemical structure of deoxynivalenol (DON).
Absorption
Intestinal absorption and metabolism of DON vary widely across animal species and depend largely on regional pH, specific intestinal segments, and bacterial activity. The localisation of the intestinal microbiota plays a significant role in determining the bioavailability of DON and its metabolites following ingestion (Knutsen et al., 2017). In addition, DON absorption varies according to animal species, age, and even sex. In most mammalian species, DON appears rapidly in the bloodstream after oral ingestion (Payros et al., 2016).
Distribution
The distribution of this toxin occurs across multiple organs, with non-uniform concentrations. Following oral ingestion, elevated levels of DON are detected in plasma, liver and kidneys within 30 minutes; subsequently, its concentration increases in the intestinal tract after one hour. This pattern can be explained by the rapid absorption of DON in the digestive tract and its brief circulation in plasma before distribution to multiple organs. DON is efficiently metabolised by hepatic enzymes and excreted via the urine, leading to reduced concentrations in plasma, liver and kidneys. In contrast, the fraction of DON that is not absorbed remains in the gastrointestinal tract and tends to accumulate, as intestinal transit is considerably slower than systemic circulation. Finally, DON has also been detected at low levels in the brain, suggesting that this toxin may cross the blood-brain barrier and potentially induce cerebral inflammation (Sun et al., 2022).
Metabolism
The metabolism of DON involves two main pathways: Phase II metabolism and intestinal biotransformation. Phase II metabolism includes the formation of glucoside, glucuronide and sulphate conjugates. In contrast, intestinal biotransformation involves the conversion of DON to deepoxi-DON (DOM-1). The relative contribution of these metabolic pathways varies among species. For example, in pigs and humans, approximately 70–94% of DON is glucuronidated, whereas only a small proportion is detoxified by the intestinal microbiota (Sun et al., 2022).
Phase II metabolism varies markedly among animal species. In birds, sulphate conjugation is the predominant pathway, whereas other species mainly rely on glucuronidation. Notably, pigs and humans share the same Phase II metabolite, DON-12-GlcA, which is why pigs are commonly used as a model species for toxicokinetic and metabolic studies relevant to humans (Sun et al., 2022).
The intestinal transformation of DON into DOM-1 by bacterial activity constitutes an important detoxification pathway. In broiler chickens, this mechanism is considered one of the reasons for their higher resistance to DON compared with other animal species. However, DOM-1 is not the only metabolite produced through biotransformation in this species, as conversion to DON-3S has also been reported (Sun et al., 2022).
Excretion
In pigs, mice, and humans, the excretion of DON and its metabolites occurs exclusively via the urinary route. In contrast, in chickens and rats, a higher proportion of DON may be excreted in faeces than in urine (Sun et al., 2022).
Zearalenone
Chemical properties
Zearalenone (ZEN) is a mycotoxin with oestrogenic activity and a resorcylic acid lactone structure. The main metabolites of zearalenone are α- and β-zearalenol, which are formed by the reduction of the ketone group to an alcohol. These metabolites are stereoisomers that differ in the chirality of the carbon atom to which the hydroxyl group is attached.
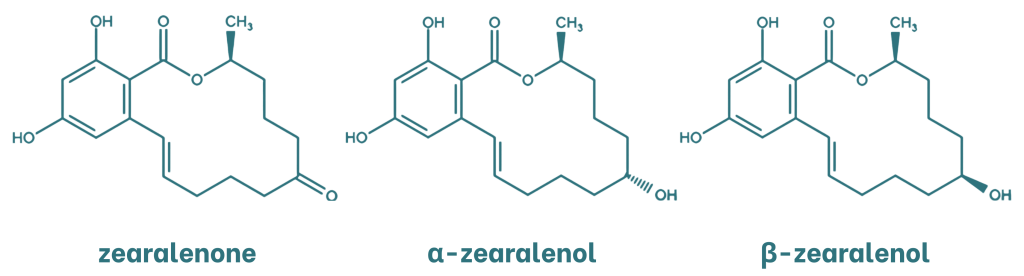
Figure 3. Chemical structure of zearalenone and its metabolites.
Absorption
ZEN is rapidly and extensively absorbed following oral administration in rats, rabbits, pigs and humans (EFSA, 2011). A distinctive feature of its absorption is that, after intestinal uptake, part of the absorbed toxin is excreted via bile and undergoes a second absorption cycle (Han et al., 2022).
Distribution
ZEN and its metabolites can be detected in animal tissues as well as in animal-derived products. The parent toxin is generally present at relatively low levels in the liver, kidneys, muscle and plasma; however, its metabolites, α- and β-zearalenol, show a wider tissue distribution. Notably, these metabolites exhibit a high affinity for albumin, which contributes to their prolonged persistence in plasma (Liu et al., 2020). The primary target organs of ZEN and its metabolites are reproductive tissues (uterus, testes and ovaries), as well as adipose tissue (Han et al., 2022).
Metabolism
The metabolism of ZEA has been extensively investigated in animals, both in vivo and through in vitro studies. Three main biotransformation pathways have been reported across different species (EFSA, 2011).
The first pathway involves enzymatic reduction catalysed by 3α- and 3β-hydroxysteroid dehydrogenases, leading to the formation of α- and β-zearalenol, as well as smaller amounts of zearalanols. These primary reduced metabolites differ in their oestrogenic activity. In mammalian species, hepatic metabolism of zearalenone varies markedly with respect to the profile of reduced and glucuronidated metabolites. For example, pigs predominantly convert zearalenone to α-zearalenol, whereas cattle mainly form β-zearalenol, which helps explain the greater sensitivity of swine to ZEN (EFSA, 2011).
The second metabolic pathway involves monohydroxylation. This process has been reported in humans and is mediated by cytochrome P450 enzymes in hepatic microsomes. Aromatic hydroxylation leads to the formation of catechols, which can subsequently be oxidised to quinones. These quinones may undergo redox cycling and covalently modify biological macromolecules. The oestrogenic properties of these catechol metabolites remain unknown (EFSA, 2011).
The final metabolic pathway involves Phase II conjugation of ZEN and its reduced metabolites with glucuronic acid and sulphate, mediated by enzymes such as uridine diphosphate-glucuronosyltransferases and sulphotransferases. Based on in vitro studies, ZEN and its metabolites are rapidly glucuronidated in the liver and intestine; they can also be metabolised in extrahepatic tissues in both humans and several animal species (EFSA, 2011).
Excretion
In most mammals, ZEN is excreted via the bile and undergoes enterohepatic circulation. The glucuronide derivatives of this mycotoxin are secreted into the bile and subsequently reabsorbed and metabolised in the intestinal mucosa. This reabsorption contributes to the retention of ZEN in the body, thereby prolonging its toxic effects in animals and hindering its elimination (Han et al., 2022).
Ochratoxin A
Chemical properties
Ochratoxins are composed of a phenylalanine moiety linked to dihydroisocoumaric acid through a peptide bond involving the carboxyl (-COOH) and amino (-NH2) groups. Two main types are recognised, ochratoxins A and B, which differ in that ochratoxin A contains a chlorine atom. Ochratoxins can undergo hydrolysis, yielding the metabolites OTα and OTβ, which exhibit lower toxicity.
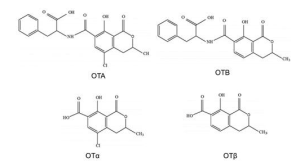
Figure 4. Chemical structure of the main ochratoxins and their metabolites (Martínez et al., 2018).
Absorption
These mycotoxins are rapidly absorbed after oral ingestion, reaching high blood levels for several hours. Ochratoxin A (OTA) enters the systemic circulation by passive diffusion through the stomach and, in particular, through the proximal region of the jejunum. This process is facilitated by the high affinity of plasma proteins for this toxin. The systemic bioavailability of the toxin varies according to the species (Schrenk et al., 2020b).
Distribution
Regarding distribution, OTA is particularly relevant due to its strong non-covalent binding to serum proteins, especially albumin, which explains its difficult elimination and long half-life in the body. OTA is distributed to all organs; however, its concentration in each organ depends on the animal species, the administered dose, and other factors. Most studies report that OTA reaches its highest concentrations in the kidneys, followed by the liver and muscle. Furthermore, redistribution and deposition of this toxin may occur in lipid-rich organs.
Regarding placental transfer, in vitro studies indicate that transport is minimal. However, available evidence suggests that OTA transfer increases as placental development progresses. In addition, OTA may be transferred via the uterine route or through maternal milk in both animals and humans. Studies in dairy cows have detected traces of OTA in milk, suggesting that the very low concentrations of this toxin in this animal product may be attributed to efficient presystemic degradation by the ruminal microbiota (Schrenk et al., 2020b).
Metabolism
The major metabolite of OTA is OTα, which is formed through hydrolysis of the amide bond between phenylalanine and dihydroisocoumaric acid. This metabolite is produced by the intestinal microbiota in non-ruminant animals, including humans, and by the ruminal microbiota in cattle, sheep and other ruminants. OTA degradation is mediated by several hydrolases, among which carboxypeptidase A appears to be the most active. The formation of OTα is considered an important detoxification process, as this metabolite does not accumulate in the kidneys and is rapidly excreted in the urine as a glucuronide (Schrenk et al., 2020b).
Excretion
The strong binding of OTA to albumin reduces its elimination by glomerular filtration. Consequently, tubular secretion—mediated by organic anion transporters—plays a major role in OTA excretion. However, the toxin can be reabsorbed at this level, a process that is partly responsible for its accumulation in the body.
In addition, several in vivo studies have shown that the toxin can be reabsorbed at any segment of the nephron via either active transport or passive diffusion, depending on pH. Biliary excretion of OTA and its metabolites also represents an elimination route. As occurs with urinary excretion, OTA may be reabsorbed after hydrolysis by the intestinal microbiota and subsequently re-enter the enterohepatic circulation, resulting in slow elimination from the body. Finally, evidence indicates that OTA can be excreted through milk, with a direct relationship between OTA intake and its concentration in milk (Kőszegi et al., 2016).
Toxin T-2
Chemical properties
T-2 toxin is a mycotoxin belonging to the type A trichothecene family. It is characterised by the presence of two ester groups at the C-4 and C-8 positions of its molecular structure. Its main metabolite is HT-2 toxin, which is formed by the hydrolysis of the ester group at the C-4 position, resulting in a hydroxyl group.
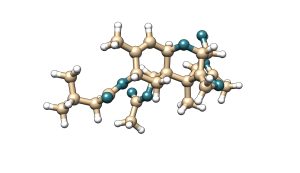
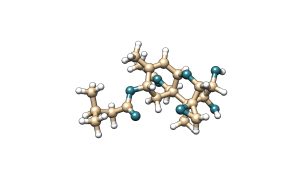
Figure 5. Chemical structure of T-2 toxin and its main metabolite HT-2.
Absorption
After ingestion of contaminated feed, T-2 toxin is rapidly absorbed in most animal species. Following absorption, it is distributed throughout the body with little or no accumulation in organs or tissues. T-2 toxin is lipophilic and, in addition to gastrointestinal absorption, can also cross the respiratory mucosa (Janik-Karpinska et al., 2022).
Distribution
T-2 toxin is rapidly transferred from plasma to tissues and organs, with a plasma half-life of less than 20 minutes. It is distributed to organs such as the liver and kidneys without significant accumulation. In addition, T-2 toxin is able to cross the placenta and reach foetal tissues (CONTAM, 2011).
Metabolism
The metabolism of T-2 toxin occurs in the intestine, liver, and other tissues (CONTAM, 2011). The main metabolic pathways involved are:
- Hydrolysis
- Hydroxylation
- Conjugation
- Deepoxidation
The predominant metabolite of T-2 toxin across all species is HT-2 toxin, which is formed through hydrolysis. This metabolite is considered an important biomarker, as its measurement allows the detection of exposure to T-2 toxin in both animals and humans (Janik-Karpinska et al., 2022). Most metabolites, and particularly T-2 toxin itself, are extensively conjugated with glucuronides, which represents one of the most relevant detoxification mechanisms (CONTAM, 2011).
Excretion
The urinary and biliary tracts are the main routes for the excretion of T-2 toxin and its metabolites. Excretion occurs rapidly, with a higher proportion eliminated in the faeces. In addition, enterohepatic recycling of T-2 toxin and its glucuronide-conjugated metabolites has been reported (CONTAM, 2011).
Fumonisins
Chemical properties
Fumonisins have a molecular structure similar to that of a lipid chain. Four types of fumonisins have been identified, which differ in the presence of alcohol groups at positions 5 and 10 of the chain. Within this group, fumonisin B1 (FB1) exhibits the highest toxicity. Additionally, these mycotoxins present ester-linked side chains at C-14 and C-15.
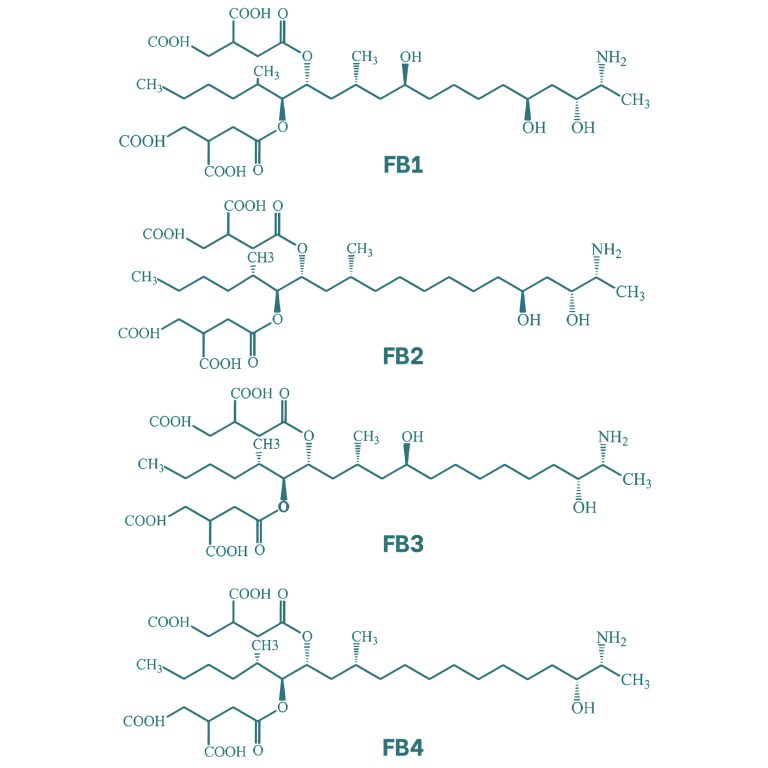
Figure 6. Chemical structure of the main fumonisins (Kostić et al., 2019).
Absorption
Fumonisins are mycotoxins characterised by low bioavailability, due to the high polarity of their structure and the limited expression of specific transport receptors in the intestine. In several species, studies indicate that fumonisins undergo a series of consecutive hydrolytic reactions, primarily in the intestine, leading to the partial formation of fumonisins A and B and, ultimately, to HFB1 (aminopentol). In exposed animals, these metabolites can be detected in the liver, kidneys and, to a lesser extent, in muscle tissue (Schrenk et al., 2022).
Distribution
After absorption in the gastrointestinal tract, FB1 is primarily distributed to the liver, kidneys, and muscle. Both the liver and kidneys are known to be particularly sensitive to this mycotoxin, which may be related to the concentrations reached in these organs following absorption (Knutsen et al., 2018).
Metabolism
The primary sites of fumonisin metabolism are the liver and the digestive tract, where the main metabolic pathways include hydrolysis, acylation, and transamination. The main metabolite of FB1 is HFB1, also known as AP1. This metabolite is formed through hydrolysis of the tricarballylic acid side chains at the C-14 and C-15 positions, which are subsequently replaced by hydroxyl groups (Wang et al., 2015).
Excretion
Most studies in farm and experimental animals report that, after oral ingestion, FB1 and FB2 are excreted unchanged via the faeces, and to a lesser extent via the urine. Excretion of these toxins occurs in the first 12 and 48 hours (Knutsen et al., 2018).
Conclusion
The toxicokinetics of mycotoxins are highly complex and vary depending on the compound and the animal species. Understanding their absorption, distribution, metabolism, and excretion (ADME) is therefore essential for designing effective strategies to minimise their toxic impact on target organs and safeguard both animal and human health.


