Fumonisins are a group of mycotoxins produced by various types of fungi, primarily from the genus Fusarium, with the most common species being F. verticillioides and F. proliferatum. These contaminants pose a significant risk to food safety and public health, as they are frequently found in staple crops such as rice, sorghum, and, most notably, maize and its derivatives (Zhou et al., 2025; Anumudu et al., 2024; Yli-Mattila & Sundheim, 2022).
Since fumonisin-producing fungi can grow over a wide range of temperatures but require high water activity, these toxins generally form before harvest or during the early stages of storage. In addition, these substances are heat-stable, making their high prevalence in feed a significant risk factor (EFSA, 2025; Schaarschmidt & Fauhl-Hassek, 2021).
Chemical characteristics of fumonisins
The B-series stands out among the various types of fumonisins identified. Within this group, fumonisin B1 (FB1), fumonisin B2 (FB2), and fumonisin B3 (FB3) have been the most extensively studied, with FB1 standing out due to its high prevalence and toxicity (EFSA, 2022; Torre-Hernández et al., 2014). From a chemical perspective, fumonisins are structurally similar diesters containing polyols and glycerol tricarboxylic acids, characterized by high polarity and water solubility (Figure 1). Their structure resembles that of sphingosine (So) and sphinganine (Sa), which is why they are classified as sphingoid-type mycotoxins. These compounds are key components of sphingomyelin, an essential lipid of cell membranes involved in fundamental processes such as proliferation, differentiation, apoptosis, senescence, and carcinogenesis, as well as in cell–cell interaction and recognition (Lassallette et al., 2025; Qu et al., 2022).
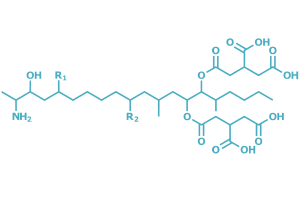
Image 1. Generic chemical structure of B-group fumonisins (Torre-Hernández et al., 2014)
The structural similarity of fumonisins to So and Sa explains their main mechanism of toxicity: the disruption of sphingolipid metabolism. In this context, the Sa:So ratio is widely used as a biomarker of fumonisin exposure across different animal species (EFSA, 2018).
Global prevalence of fumonisins: a real challenge
In recent years, various studies have confirmed that fumonisin contamination in cereals and feed represents a global challenge. Farhadi et al. (2021) conducted a systematic review and meta-analysis of thousands of cereal-based food samples collected between 1990 and 2020, identifying a significant prevalence of fumonisins across multiple regions, with particularly high concentrations in maize-derived products. Consistent with these findings, the ten-year global study published by Gruber-Dorninger et al. (2019) revealed that 88% of analyzed feeds contained at least one mycotoxin, with fumonisins showing a high prevalence (60%) and particularly affecting maize, which exhibited an incidence of 80%.
Similarly, global mycotoxin surveys report fumonisin presence in over 60–70% of samples analyzed, both in raw materials and compound animal feeds, with variations depending on geographic region and climatic factors (Lopes et al., 2023; Akello et al., 2021; Cheng et al., 2002).
Toxicokinetics and toxicity of fumonisins
Regarding their toxicokinetics, fumonisins generally exhibit low bioavailability, mainly due to their charged structure (with three negative charges) and the likely low expression of specific intestinal transporters (EFSA, 2018; Shier, 2000). However, despite this limited absorption, these contaminants and their metabolites can induce systemic toxicity, with both clinical and subclinical effects.
In terms of metabolism, although qualitative and quantitative differences exist between animal species, fumonisins typically undergo sequential hydrolytic reactions primarily in the gastrointestinal tract, initially forming partially hydrolyzed fumonisins A and B (pHF1a and pHF1b), and subsequently HFB₁ (also known as aminopentol, AP). In exposed animals, both the parent compound and its metabolites can be detected in the liver and kidneys, and to a lesser extent in muscle tissue (Anumudu et al., 2024; Qu et al., 2022).
Excretion of these toxins occurs predominantly via feces, with urinary excretion being considerably lower (Knutsen et al., 2018; Hartinger et al., 2011; Guerre et al., 2022). Furthermore, carry-over of fumonisins has been reported in animal-derived products such as eggs and meat (Osaili et al., 2022; Dazuk et al., 2020).
The toxicity of fumonisins is primarily based on the inhibition of ceramide synthase, which disrupts sphingolipid biosynthesis and leads to sphinganine accumulation, impairing cell growth and nervous tissue signaling. This mechanism is compounded by the induction of oxidative stress and cytotoxicity, resulting in severe tissue damage and hepatotoxicity. Consequently, these mycotoxins cause immunosuppression and trigger gastrointestinal issues, ultimately leading to reduced performance (Qu et al., 2022; Kulcsár et al., 2023).
Sensitivity to the effects of fumonisins varies considerably between species. While horses and pigs are highly susceptible and can develop specific syndromes such as equine leukoencephalomalacia and porcine pulmonary edema, poultry exhibit relatively greater resistance. Nevertheless, chronic exposure affects their performance, intestinal health, and immune response. Furthermore, the health risk is exacerbated by the frequent co-occurrence of fumonisins with other mycotoxins, such as deoxynivalenol, zearalenone, and aflatoxins in feed, leading to synergistic or additive interactions that enhance toxicity, as evidenced by increased liver lesions and carcinogenic effects when combined with aflatoxin B1 (Anumudu et al., 2024; Guerre et al., 2022; Qu et al., 2022).
BIŌNTE® QUIMITŌX® PLUS: fumonisin mitigation strategy
In this context, BIŌNTE® QUIMITŌX® PLUS emerges as an effective strategy against fumonisins, featuring a formula based on three modes of action: adsorption, bioprotection, and postbiotic effects. Firstly, the formulation includes a blend of high-quality selected clays, highly effective in fumonisin adsorption. These clays act through two types of interactions: polar interactions with surface cations and the formation of organometallic complexes (mycotoxin–clay) with structural magnesium centers.
Additionally, BIŌNTE® QUIMITŌX® PLUS contains two key phytogenic ingredients that mitigate fumonisin toxicity: curcumin and silymarin. Curcumin mitigates cellular toxicity by reducing sphingolipid accumulation and restoring intracellular calcium balance disrupted by fumonisins, and has been shown to possess potent hepatoprotective and antioxidant effects. Complementarily, silymarin acts as a direct cytoprotectant, significantly reducing hepatocyte apoptosis through a molecular mechanism involving regulation of the inflammatory response and modulation of liver tissue damage (He et al., 2004; Ledur et al., 2020).
Finally, BIŌNTE® QUIMITŌX® PLUS includes hydrolyzed yeast and yeast cell wall components. The β-glucans and mannan-oligosaccharides present in the cell wall contribute to mycotoxin binding in the gastrointestinal tract (Compagnollo et al., 2020; Elnaggar et al., 2014). These ingredients also strengthen the intestinal barrier and promote the growth of beneficial gut bacteria, enhancing digestion, nutrient absorption, and microbial degradation of fumonisins. Moreover, β-glucans have been shown to exert an immunomodulatory effect, reinforcing the immune response against infections and the stress induced by mycotoxins (Patterson et al., 2023; Pascual et al., 2020).
BIŌNTE® QUIMITŌX® PLUS demonstrates its efficacy under in vitro and in vivo conditions
This triple-action solution has demonstrated its efficacy under in vitro conditions in studies conducted by the Trilogy Laboratory in the United States. BIŌNTE® QUIMITŌX® PLUS showed high efficacy in the in vitro adsorption of a broad range of mycotoxins, including emerging and modified mycotoxins. Specifically, for fumonisins, the product achieved over 78% adsorption for FB1 and approximately 90% for FB2 and FB3, with a minimal desorption rate of around 0.2% in all cases. These in vitro results have been further supported by in vivo trials conducted across various terrestrial species.
Efficacy of BIŌNTE® QUIMITŌX® PLUS against fumonisins in poultry
Firstly, a toxicokinetic study was conducted in poultry in collaboration with Ghent University (Belgium) to evaluate the FB1 adsorption capacity in these animals. In broiler chickens, the absorption of this mycotoxin occurs rapidly in the proximal portion of the small intestine, making it detectable in the bloodstream shortly after ingestion of contaminated feed (Antonissen et al., 2020). Therefore, it is essential to ensure a rapid action of anti-mycotoxin products to prevent FB1 from entering the bloodstream and, consequently, its systemic distribution and toxicity.
In this study, BIŌNTE® QUIMITŌX® PLUS demonstrated high efficacy just 5 minutes after administration. Supplementation resulted in a 74% reduction in the maximum plasma concentration of FB1 detected in the animals, as well as an 82% decrease in the overall bioavailability of this mycotoxin (Figure 1). These results confirm that the product significantly reduces the entry of the mycotoxin and its metabolites into systemic circulation, thereby protecting animal health.
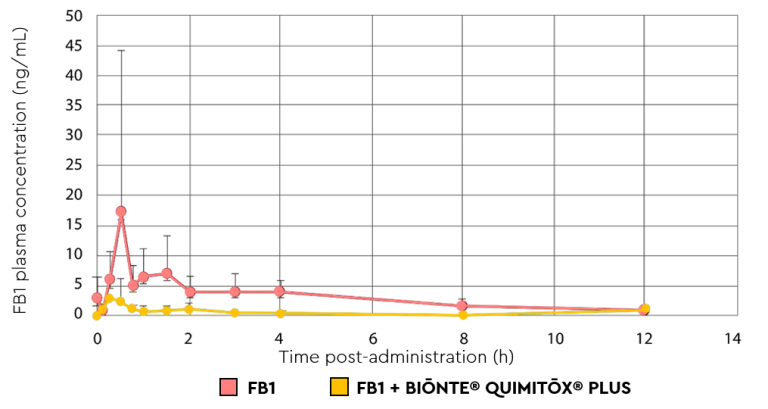
Figure 1. Plasma concentration profile of FB1 over time following oral administration of a 2.5 mg FB1/kg bolus with and without BIŌNTE® QUIMITŌX® PLUS (3 g/kg body weight).
Following this evaluation of BIŌNTE® QUIMITŌX® PLUS efficacy, an in vivo trial conducted in collaboration with the Samitec Institute and the University of Santa Maria (Brazil) monitored the sphinganine/sphingosine ratio, a recognized biomarker of fumonisin exposure, in broiler chickens exposed to FB1. In this study, a 3.8% reduction in this reference parameter was observed, demonstrating that the product is highly effective against fumonisins (EFSA, 2010, 2018; Guerre et al., 2022).
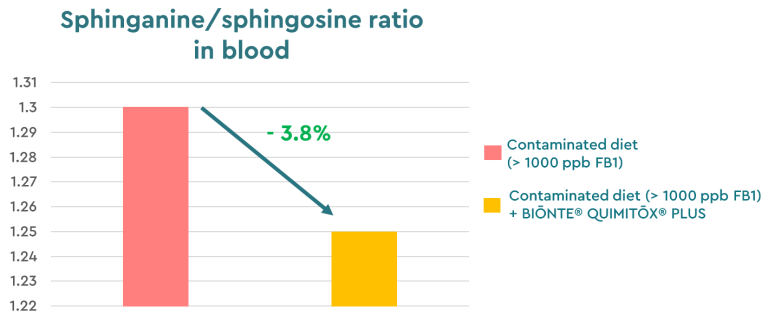
Figure 2. Sphinganine/sphingosine ratio in broilers exposed to FB1 and FB1 + BIŌNTE® QUIMITŌX® PLUS.
Within this same trial, BIŌNTE® QUIMITŌX® PLUS improved the productive performance of broiler chickens fed a contaminated diet. Increases were observed in body weight (+0.7%), average daily gain (+2.9%), and intestinal absorptive surface (+14.6%), all parameters negatively affected by fumonisin exposure. Additionally, the product reduced the relative liver weight of the animals (−6.1%), indicating mitigation of the inflammatory effects of these mycotoxins (+12.9%), thereby helping to maintain liver integrity in broilers exposed to FB1 (Figure 3).
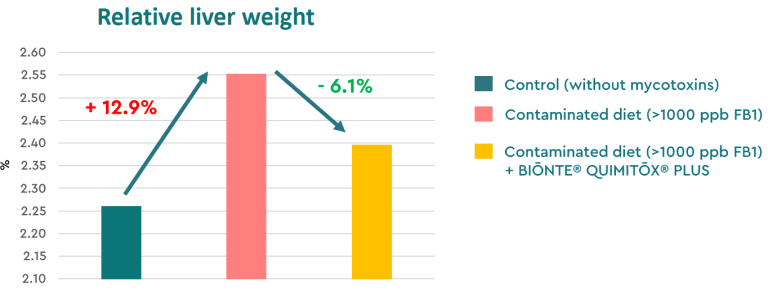
Figure 3. Relative liver weight in broiler chickens: unexposed to mycotoxins, exposed to FB1, and exposed to FB1 + BIŌNTE® QUIMITŌX® PLUS.
Furthermore, the efficacy of BIŌNTE® QUIMITŌX® PLUS in laying hens exposed to fumonisins was evaluated, given the importance of mycotoxin effects in this production category. A field study was conducted in Mexico on a group of hens fed a diet naturally contaminated with fumonisins, as well as AFB1 and deoxynivalenol (DON). The trial demonstrated significant improvements in productive performance, including a reduction in feed conversion ratio (−5.8%) and an increase in the percentage of commercially acceptable eggs (+2.7%). Additionally, a 0.25% decrease in mortality was observed, along with improvements in liver health confirmed through necropsy.
Efficacy of BIŌNTE® QUIMITŌX® PLUS against fumonisins in swine
Swine are highly sensitive to fumonisins, often developing specific respiratory pathologies. The efficacy of BIŌNTE® QUIMITŌX® PLUS has therefore been demonstrated in this species through various studies focused on weaned piglets and breeding sows, in collaboration with the University of Thessaly (Greece).
One study involving weaned piglets used a diet naturally contaminated with FB1 and FB2. The results showed increases in body weight (+19.5%) and feed intake (+24.4%), along with a significant reduction in mortality, attributable to improved immune response and overall health in these young animals, which are highly susceptible to secondary diseases. In this context, BIŌNTE® QUIMITŌX® PLUS also demonstrated its ability to reduce oxidative stress biomarkers (Figure 4), and histopathological analyses revealed its capacity to counteract fumonisin-induced lesions in the liver (degeneration and infiltration) and intestinal epithelium (reduction of nutritional absorptive surface).
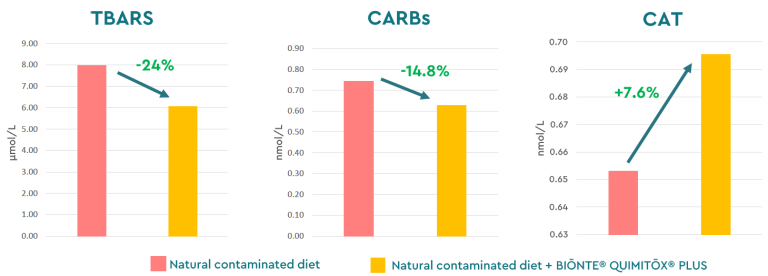
Figure 4. Oxidative stress biomarkers in piglets exposed to fumonisins (FB1, FB2) with and without BIŌNTE® QUIMITŌX® PLUS. *Thiobarbituric Acid Reactive Substances (TBARS), Protein Carbolines (CARBs) and Total Antioxidant Capacity (TAC).
It should be added that these results were confirmed in a second in vivo study conducted in piglets, this time exposed to fumonisins in combination with aflatoxin B1 (AFB1). Again, BIŌNTE® QUIMITŌX® PLUS demonstrated its ability to improve productive parameters, organ health, and antioxidant status in animals fed this co-contaminated diet.
Regarding the efficacy of BIŌNTE® QUIMITŌX® PLUS against fumonisins in sows, two in vivo studies were conducted with the University of Thessaly (Greece) under multi-contamination conditions. In these studies, the product demonstrated protective effects in sows exposed to FB1, FB2, zearalenone (ZEN), and T-2 toxin, as well as in sows challenged with FB1, FB2, and AFB1. In both cases, BIŌNTE® QUIMITŌX® PLUS reduced oxidative stress caused by the mycotoxins and improved mammary gland health, alleviating inflammation symptoms (Figure 5).
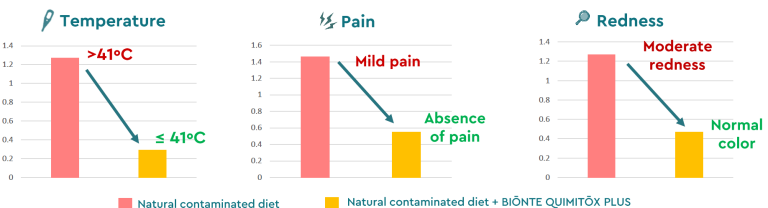
Figure 5. Inflammation biomarkers (temperature, pain, and redness) in gestating sows exposed to a multi-contaminated diet (FB1, FB2, AFB1) with and without BIŌNTE® QUIMITŌX® PLUS.
These trials in sows also included litter characteristics, reporting in both cases a reduction in the total number of stillborn and mummified piglets per litter, and an increase in the number of weaned piglets, thanks to the administration of BIŌNTE® QUIMITŌX® PLUS (Figure 6).
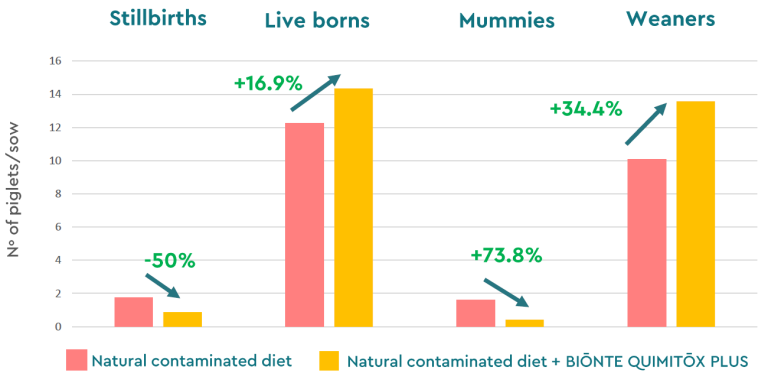
Figure 6. Litter characteristics in sows exposed to a multi-contaminated diet (FB1, FB2, ZEN, T-2) with and without BIŌNTE® QUIMITŌX® PLUS.
Efficacy of BIŌNTE® QUIMITŌX® PLUS against fumonisins in ruminants
Regarding the effectiveness of BIŌNTE® QUIMITŌX® PLUS against fumonisins in ruminants, an in vitro trial was conducted in ruminal fluid in collaboration with the Catholic University of the Sacro Coure (Italy). The study demonstrated the ability of BIŌNTE® QUIMITŌX® PLUS to mitigate the harmful effects of these mycotoxins in dairy cows, achieving a 63% reduction in fumonisin concentration in the ruminal fluid of treated cows (Figure 7).
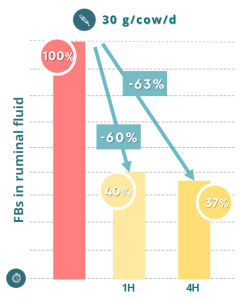
Figure 7. Reduction of fumonisins (FBs) concentration in ruminal fluid with BIŌNTE® QUIMITŌX® PLUS.
Finally, BIŌNTE® QUIMITŌX® PLUS has also demonstrated its efficacy against fumonisins in dairy sheep under co-contamination conditions with AFB1 and ochratoxin A (OTA) during the peripartum and lactation periods. In this study, conducted in collaboration with the University of Thessaly (Greece), supplementation resulted in a 44% reduction in the incidence of postpartum fever and a 34% decrease in mastitis cases (Figure 8).
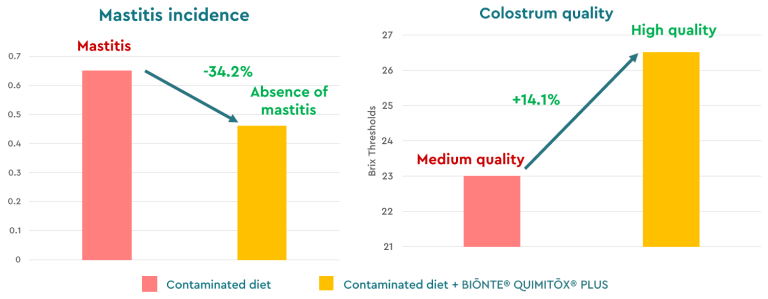
Figure 8. Incidence of mastitis and colostrum quality in dairy sheep exposed to a multi-contaminated diet (FB1, FB2, ZEN, T-2) with and without BIŌNTE® QUIMITŌX® PLUS.
Regarding productivity, a significant improvement in milk quality was observed, with a 10% increase in fat content and a 1.5% increase in protein. Colostrum quality improved by 14% (Brix values), ensuring better neonatal immunity (Figure 8). Additionally, the product demonstrated significant hepatoprotective effects, reducing liver damage, indicating enzymes such as ALT (−73%) and AST (−32.4%) (Figure 9).
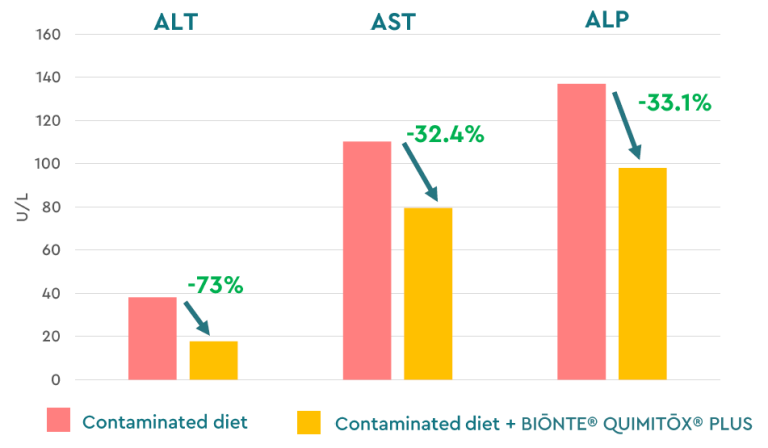
Figure 9. Liver damage biomarkers in dairy sheep exposed to a multi-contaminated diet (FB1, FB2, ZEN, T-2) with and without BIŌNTE® QUIMITŌX® PLUS. * Alanine Aminotransferase (ALT), Aspartate Aminotransferase (AST), and Alkaline Phosphatase (ALP).
Conclusion
In this way, BIŌNTE® QUIMITŌX® PLUS has demonstrated its effectiveness against fumonisins, a group of mycotoxins that are highly prevalent worldwide and known for their negative effects on the health and performance of various animal species. These mycotoxins, commonly found in cereals and plant-based feed by-products, can affect key organs such as the liver, immune system, and digestive tract, leading to significant production losses.
In vitro and in vivo studies showed that supplementation with BIŌNTE® QUIMITŌX® PLUS not only mitigated the toxic effects of fumonisins but also improved productive parameters while supporting liver and intestinal health. These results confirm that BIŌNTE® QUIMITŌX® PLUS is a reliable tool to protect animal health and optimize performance in the presence of fumonisins.


