Intestinal barrier: the first line of biological defense
The gastrointestinal tract is not only the central axis for digestion and nutrient absorption, it also constitutes the first physiological barrier against feed-borne contaminants (Assunçao et al., 2020; Alassane-Kpembi et al., 2019). Currently, the challenge posed by mycotoxins is global and persistent, with estimates suggesting that these secondary fungal metabolites contaminate between 60% and 80% of crops worldwide (Gasperini et al., 2025; Guerre, 2020).
Once ingested, these toxins reach the intestinal lumen, where they exert a direct impact on mucosal integrity. This process simultaneously compromises epithelial cell viability, mucus layer functionality, and the stability of the microbial ecosystem, disrupting the homeostasis required for efficient and healthy animal production (Gasperini et al., 2025; Celi et al., 2017).
Intestinal permeability: a selective barrier
The structural and functional integrity of the intestinal barrier is intrinsically dependent on tight junctions, highly specialized protein complexes that hold the lateral membranes of enterocytes together (Figure 1). These complexes act as a dynamic seal between adjacent cells, establishing a continuous protective barrier whose fundamental mission is the selective regulation of molecular flow from the lumen into the bloodstream (Gasperini et al., 2025; Zhai et al., 2021).
The functionality of tight junctions is based on a precise interaction between various protein groups that dictate barrier stability. In this framework, transmembrane proteins, such as claudins (1, 3, and 4), occludin, tricellulin, and junctional adhesion molecules (JAMs), project into the intercellular space to consolidate a robust physical seal. However, the efficacy of this complex directly depends on cytoplasmic scaffolding proteins, most notably ZO-1, which links transmembrane proteins to the actin cytoskeleton to ensure mechanical stability and the dynamic regulation of the paracellular pore (Gao et al., 2018; Huang et al., 2022; Zhai et al., 2021).
Under optimal physiological conditions, tight junctions act as a selective filter that promotes the absorption of nutrients and electrolytes while restricting the passage of antigens and pathogens into the bloodstream (Gasperini et al., 2025; Zhai et al., 2021). In technical terms, epithelial integrity is monitored through transepithelial electrical resistance (TEER), thus, a reduction in this parameter is an unequivocal indicator that the barrier has been compromised (Gao et al., 2018; Huang et al., 2022; Zhai et al., 2021).
Mycotoxins: the collapse of tight junctions
Epithelial permeability can be affected by multiple factors, including mycotoxins. Within these chemically diverse metabolites, the toxic pathways through which they alter structural integrity vary. On one hand, some of the most prevalent mycotoxins, such as deoxynivalenol (DON) and ochratoxin A (OTA), are notable for their ability to drastically reduce TEER and, consequently, increase intestinal permeability (Gao et al., 2018; Antonissen et al., 2014). While DON exerts its toxicity by inhibiting protein synthesis at the ribosomal level, specifically affecting the expression of claudin-4, OTA intervenes by altering the fundamental structure of occludin (Gasperini et al., 2025; Pierron et al., 2016).
On the other hand, zearalenone (ZEN) induces differential mechanical damage through the activation of the RhoA/ROCK signaling pathway. This mechanism triggers a remodeling of the actin cytoskeleton that severely alters epithelial permeability (Huang et al., 2022). Complementarily, T-2 and HT-2 toxins act on the gene expression of claudin-1, occludin, and ZO-1, precipitating a loss of cellular adhesion that serves as a gateway for various enteric pathogens (He et al., 2023).
Similarly, even at concentrations that could be considered low, aflatoxins (AFB1 and AFM1) have been shown to alter the distribution of tight junctions (Gao et al., 2018; Gasperini et al., 2025). Finally, fumonisins exhibit a unique mechanism of action: by inhibiting ceramide synthase, they alter the composition of membrane sphingolipids, compromising the formation and maintenance of tight junctions directly from the cell’s lipid structure (Bergen et al., 2025; Gasperini et al., 2025).
| Mycotoxin | Primary mechanism of action | Proteins/pathways affected | Functional consequence |
|---|---|---|---|
| Deoxynivalenol (DON) | Inhibition of protein synthesis at ribosomal level | Claudin-4 expression | Drastic reduction in TEER and increase in paracellular flow |
| Ochratoxin A (OTA) | Alteration of fundamental protein structure | Occludin structure | Drastic reduction in TEER and increase in paracellular flow |
| Zearalenone (ZEN) | Activation of a cellular signaling pathway | RhoA/ROCK signaling pathway, which remodels the actin cytoskeleton | Severe compromise of epithelial permeability |
| T-2 and HT-2 toxins | Alteration of gene expression | Claudin-1, occludin, and ZO-1 expression | Loss of cellular adhesion, creating a gateway for enteric pathogens |
| Aflatoxins (AFB1, AFM1) | Modification of protein localization | Tight junction distribution | Barrier alteration even at low concentrations |
| Fumonisins | Inhibition of membrane lipid synthesis | Ceramide synthase, affecting sphingolipid composition | Compromise of the formation and maintenance of tight junctions |
Table 1. Summary of the effects of major mycotoxins on intestinal barrier integrity.
This set of aggressions converges in the phenomenon of ‘leaky gut’ (Figure 1). The breakdown of this barrier allows for the massive translocation of luminal antigens and bacteria into the systemic circulation, a process that not only weakens the animal’s health status but also triggers chronic inflammatory responses with a high metabolic cost (Gasperini et al., 2025; Smith et al., 2012).
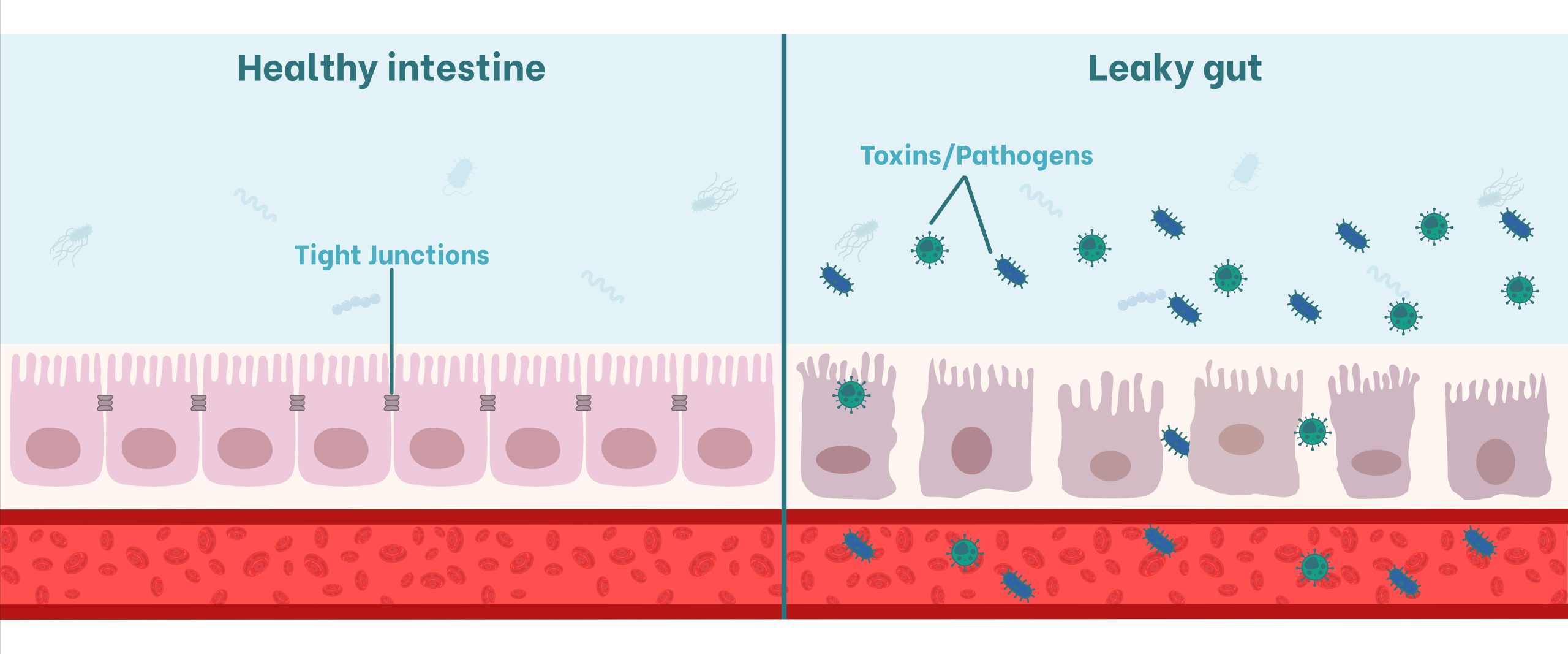
Figure 1. Representation of the impact of mycotoxins on the intestinal barrier. On the left, a healthy epithelium with functional tight junctions; on the right, the leaky gut phenomenon, where the degradation of transmembrane and/or cytoplasmic scaffolding proteins allows for the translocation of toxins and pathogens into the bloodstream.
Microbiota: a strategic axis for homeostasis
Beyond being a resident population, the intestinal microbiota should be considered a dynamic «metabolic organ» that maintains a symbiotic balance with the host (Alassane-Kpembi et al., 2019). The microbiota is fundamental for regulating intestinal mucosal integrity, as well as for ensuring proper immune function and guaranteeing optimal nutrient absorption and metabolism (An et al., 2025; Celi et al., 2017). Its functions can be classified into three pillars: protective, structural, and metabolic.
Firstly, the microbiota performs protective functions by establishing a competitive biological barrier. This mechanism prevents pathogen colonization through the displacement of invading microorganisms, competition for receptor sites, and the active production of antimicrobial factors (Celi et al., 2017; Guerre, 2020).
On the other hand, its structural functions are vital for the fortification of the gastrointestinal barrier, inducing the synthesis of immunoglobulin A (IgA) and promoting the apical sealing of tight junctions (Celi et al., 2017).
Finally, regarding the metabolic functions of the microbiota, it enables the fermentation of non-digestible dietary residues to produce short-chain fatty acids (SCFAs). These metabolites not only represent a primary cellular energy source for the epithelium but are also pillars of colonic health and the synthesis of essential vitamins, enzymes, and amino acids (Celi et al., 2017; Zheng et al., 2018). Within the metabolic functions, the defense role must also be highlighted, the microbiota is capable of hydrolyzing or reducing toxins into significantly less toxic metabolites before their systemic absorption (Guerre, 2020; Shanmugasundaram et al., 2023). This biotransformation capacity, coupled with its involvement in communication through the microbiota-gut-brain axis, positions the microbial ecosystem as the guardian of the animal’s systemic homeostasis (Celi et al., 2017; Guerre, 2020).
Mycotoxins: the imbalance of the microbial ecosystem
Mycotoxins act as potent disrupting agents of microbiota balance (Figure 2), inducing states of dysbiosis through direct antimicrobial properties or, indirectly, through cellular toxicity and the release of antimicrobial peptides that alter the intestinal ecological niche (Guerre, 2020; Gasperini et al., 2025).
Exposure to AFB1 leads to a reduction in phylogenetic diversity, altering the relationship between the main phyla and promoting a dominance of Firmicutes over Actinobacteria (Gasperini et al., 2025; Guerre, 2020). For its part, DON exerts selective pressure that modifies taxonomic abundance, this process reduces Proteobacteria populations while favoring the growth of pro-inflammatory bacteria such as Enterobacteriaceae, thereby increasing the sanitary challenge (Shanmugasundaram et al., 2023; Gasperini et al., 2025).
Similarly, ZEN negatively impacts global diversity, with a critical decrease in beneficial bacteria observed in key segments such as the cecum and colon in rabbits, pigs, and mice (Guerre, 2020; Gasperini et al., 2025). In the case of OTA, its presence increases the relative abundance of the genus Bacteroides, linked to systemic inflammation when present in high proportions, and drastically reduces butyric acid-producing populations, thereby compromising the main energy substrate for colonocytes (Zhai et al., 2021). Finally, fumonisins (FB1 and FB2) interfere with the natural succession of bacterial species, promoting the proliferation of genera that compromise gastrointestinal tract stability from early stages (Guerre, 2020; Alassane-Kpembi et al., 2019).
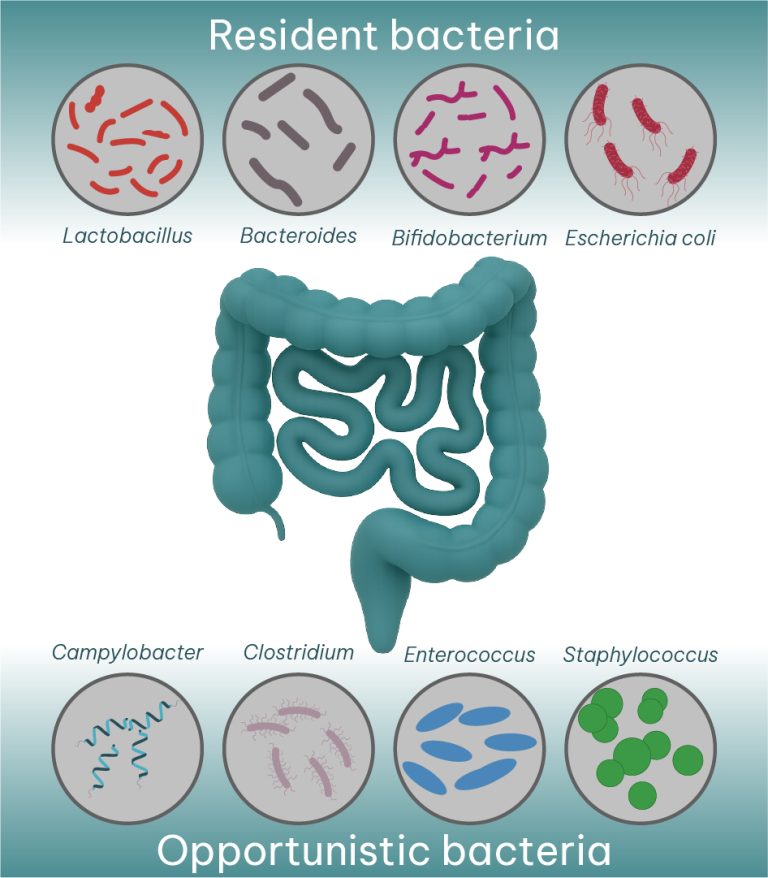
Figure 2. Microbiota composition is balanced between resident bacterial genera (e.g., Lactobacillus) and opportunistic or potentially pathogenic ones (e.g., Clostridium). Mycotoxins cause an imbalance by decreasing beneficial bacterial populations and increasing pathogenic ones.
Clinical and productive consequences
All the described alterations produced by the various mycotoxins at the intestinal level, which may involve the disruption of epithelial integrity and dysbiosis of the bacterial population, have a direct impact on animal health, and consequently, on performance.
The leaky gut phenomenon facilitates the bacterial translocation of systemic pathogens such as Salmonella Typhimurium and Escherichia coli. These microorganisms succeed in breaching the intestinal barrier to colonize mesenteric lymph nodes and vital organs, such as the spleen, liver, and lungs, triggering a chronic systemic inflammatory response and the suppression of the specific immune response (Guerre, 2020; Pierron et al., 2016).
Likewise, an increase in the bacterial translocation of pathogens increases the animals’ predisposition to secondary enteric infections. In broilers, mycotoxins such as DON and FB1 act as predisposing factors in the pathogenesis of necrotic enteritis caused by Clostridium perfringens (Antonissen et al., 2014; Shanmugasundaram et al., 2023; Guerre, 2020).
On the other hand, T-2 toxin represents a specific threat by inducing a profound ileal dysbiosis that elevates serum lipopolysaccharide (LPS) levels. This event activates hepatic inflammatory cascades in pigs, promoting pathologies such as steatosis and cellular infiltration, which compromises the animal’s basal metabolism (An et al., 2025; He et al., 2024).
For its part, DON exerts an inhibitory effect on critical transporters such as SGLT-1, which is responsible for the transport of glucose and sodium. This interference not only causes malabsorption of sugars but also triggers chronic diarrhea by compromising water reabsorption (Pierron et al., 2016; Smith et al., 2012).
This cascade of events has a decisive impact on productive parameters. The co-exposure to fumonisins and DON, even at sub-clinical doses, significantly reduces body weight gain and inefficiently elevates the feed conversion ratio (Shanmugasundaram et al., 2023; Pierron et al., 2016).
Finally, the clinical picture is worsened by the induction of anorexia and feed refusal. This is because mycotoxins such as DON and T-2 toxin abnormally stimulate satiety hormones and various cytokines that alter communication within the microbiota-gut-brain axis. This neuroendocrine imbalance drastically limits energy intake, consolidating a growth retardation that compromises the economic viability of the farm (Guerre, 2020; Smith et al., 2012; An et al., 2025).
From a strictly productive perspective, mycotoxin-induced intestinal damage translates into a direct decrease in farm profitability. The impact begins with a severe impairment of nutritional absorption function; exposure to these metabolites causes histomorphological alterations that include the shedding and atrophy of villi, drastically reducing the useful surface area for nutrient uptake (Zhai et al., 2021; Pierron et al., 2016).
Conclusion
In conclusion, the control of digestive health, through the maintenance of intestinal integrity and permeability, alongside the prevention of dysbiosis, constitutes one of the central pillars in mitigating mycotoxin-induced toxicity. In this context, a thorough understanding of the mechanisms by which these toxins interact with the gastrointestinal tract is essential for designing effective nutritional strategies capable of preserving gut health, minimizing the pathophysiological impact of mycotoxins, and, consequently, optimizing productive performance.


